Difference between revisions of "MAG"
m |
m |
||
| Line 40: | Line 40: | ||
<br> | <br> | ||
=== Cellular expression of GBP and ligands === | === Cellular expression of GBP and ligands === | ||
| − | Myelinating cells namely oligodendrocytes or Schwann cells <br> | + | Myelinating cells, namely oligodendrocytes or Schwann cells, express MAG.<br> |
=== Biosynthesis of ligands === | === Biosynthesis of ligands === | ||
<br> | <br> | ||
Revision as of 21:59, 21 June 2010
Myelin-associated glycoprotein (MAG, Siglec-4) is unique among the siglecs in that it is expressed exclusively on neuronal glial cells[1][2]. It is the most highly conserved among the siglecs in mammalian species. This siglec paradigm is unique in its activity for stabilizing axon-myelin interactions. MAG has a cytoplasmic domain that is devoid of ITIMs, but contains a tyrosine-based motif associated with binding the FYN tyrosine kinase, believed to play a role in its activity in myelin-axon interactions. MAG recognizes as ligands sialoside sequences found on gangliosides that are abundant in axonal membranes[2]. It is one of several proteins in myelin that negatively regulate axon outgrowth following tissue injury, an activity that involves MAG ligand interactions. Evidence suggests that inhibition of MAG ligand interactions may enhance neurite outgrowth and repair of injured neurons[3][4].
CFG Participating Investigators contributing to the understanding of this paradigm
Several CFG Participating Investigators (PIs) have contributed to identification of MAG as a siglec and to understanding the functions of MAG, including: Paul Crocker, Sørge Kelm, James Paulson, Ronald Schnaar
Progress toward understanding this GBP paradigm
Carbohydrate ligands
The glycan specificity of Siglec-4 has been investigated using resialylated erythrocytes[5], gangliosides[6][7], inhibition assays with oligosaccharides[8][9].
determinant recognized:
on glycolipids and/or glycoproteins
specificity for linkage of sialic acid to underlying Gal
about 10-fold better binding to Neu5Acα2,3Gal-R than Neu5Acα2,6Gal
underlying glycan structures can enhance binding
similar binding to the following structures
enhanced binding through additional internal sialic acids
higher binding to
Cellular expression of GBP and ligands
Myelinating cells, namely oligodendrocytes or Schwann cells, express MAG.
Biosynthesis of ligands
Structure
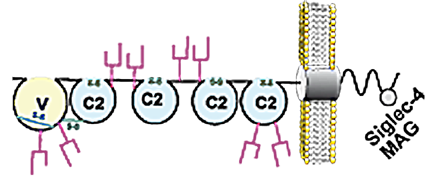
Siglec-4 is a heavily glycosylated protein of about 100kDa with 30% of its mass being made up by carbohydrates distributed over eight glycosylation sites. The extracellular part of Siglec-4 consists of five Ig-like domains (one V-set domain and four C2-set domains). Two splice variants for Siglec-4 are found in mammals, L-MAG (72kDa) and S-MAG (67kDa), which differ in their cytoplasmic domain. Whereas L-MAG contais a tyrosine phosphorylation site[10][11], which is missing in S-MAG.
Biological roles of GBP-ligand interaction
CFG resources used in investigations
The best examples of CFG contributions to this paradigm are described below, with links to specific data sets. For a complete list of CFG data and resources relating to this paradigm, see the CFG database search results for Siglec-4.
Glycan profiling
Glycogene microarray
Knockout mouse lines
The CFG has phenotyped the MAG-deficient mouse.
Glycan array
Investigators have used CFG carbohydrate compounds to study MAG ligand specificity.
Related GBPs
Compared to other Siglecs, Siglec-4 is most conserved. Based on sequence similarity orthologous proteins can be identified in all vertebrate genomes available so far (several mammals, chicken, Xenopus, zebrafish and fugu). Sialic acid binding activity selective for 2,3-linked Sia has been shown for the avian ortholog (SMP[12]) and fish Siglec-4 from zebrafish (Danio rerio) and fugu (Takifugu rubripes)[13]). Whereas the primary sequences of the Sia-binding N-terminal domains is 97 % identical between rodents and man and share over 50 % sequence identity between fish and mammals, the cytoplasmic tail is much less conserved (20% identical amino acids between fish and mammals[14]).
References
- ↑ Crocker, P. R., Paulson, J. C. & Varki, A. Siglecs and their roles in the immune system. Nat Rev Immunol 7, 255-266 (2007).
- ↑ 2.0 2.1 Schnaar, R. L. Brain gangliosides in axon-myelin stability and axon regeneration. FEBS Lett (2009).
- ↑ Yang, L. J. et al. Sialidase enhances spinal axon outgrowth in vivo. Proc Natl Acad Sci U S A 103, 11057-11062 (2006).
- ↑ Vyas, A. A., Blixt, O., Paulson, J. C. & Schnaar, R. L. Potent glycan inhibitors of myelin-associated glycoprotein enhance axon outgrowth in vitro. J Biol Chem 280, 16305-16310 (2005).
- ↑ Kelm, S. et al. Sialoadhesin, myelin-associated glycoprotein and CD22 define a new family of sialic acid-dependent adhesion molecules of the immunoglobulin superfamily. Curr Biol. 4, 965-972 (1994)
- ↑ Collins, B. E., Kiso, M., Hasegawa, A., Tropak, M. B., Roder, J. C., Crocker, P. R., Schnaar, R. L. Binding specificities of the sialoadhesin family of I-type lectins. Sialic acid linkage and substructure requirements for binding of myelin-associated glycoprotein, Schwann cell myelin protein, and sialoadhesin. J Biol Chem. 272, 16889-16895 (1997)
- ↑ Collins, B. E., Yang, L. J., Mukhopadhyay, G., Filbin, M. T., Kiso, M., Hasegawa, A., Schnaar, R.L. Sialic acid specificity of myelin-associated glycoprotein binding. J Biol Chem. 272, 1248-1255 (1997)
- ↑ Strenge, K., Schauer, R., Bovin, N., Hasegawa, A., Ishida, H., Kiso, M., Kelm, S. Glycan specificity of myelin-associated glycoprotein and sialoadhesin deduced from interactions with synthetic oligosaccharides. Eur J Biochem. 258, 677-685 (1998)
- ↑ Blixt, O., Collins, B. E., van den Nieuwenhof, I. M., Crocker, P. R., Paulson, J. C. Sialoside specificity of the siglec family assessed using novel multivalent probes: identification of potent inhibitors of myelin-associated glycoprotein. J Biol Chem. 278, 31007-31019 (2003)
- ↑ Umemori, H., Sato, S., Yagi, T., Aizawa, S., Yamamoto, T. Initial events of myelination involve Fyn tyrosine kinase signalling. Nature 367, 572-576 (1994)
- ↑ Jaramillo, M. L., Afar, D. E., Almazan, G., Bell, J. C. Identification of tyrosine 620 as the major phosphorylation site of myelin-associated glycoprotein and its implication in interacting with signaling molecules. J Biol Chem. 269, 27240-27245 (1994)
- ↑ Collins, B.E., Ito, H., Sawada, N., Ishida, H., Kiso, M., Schnaar, R.L. Enhanced binding of the neural siglecs, myelin-associated glycoprotein and Schwann cell myelin protein, to Chol-1 (alpha-series) gangliosides and novel sulfated Chol-1 analogs. J Biol Chem. 274, 37637-37643 (1999)
- ↑ Lehmann, F., Gäthje, H., Kelm, S., Dietz, F. Evolution of sialic acid-binding proteins: molecular cloning and expression of fish siglec-4. Glycobiology 14, 959-968 (2004)
- ↑ Lehmann, F., Gäthje, H., Kelm, S., Dietz, F. Evolution of sialic acid-binding proteins: molecular cloning and expression of fish siglec-4. Glycobiology 14, 959-968 (2004)
Acknowledgements
The CFG is grateful to the following PIs for their contributions to this wiki page: Paul Crocker, Sorge Kelm, James Paulson, Ron Schnaar